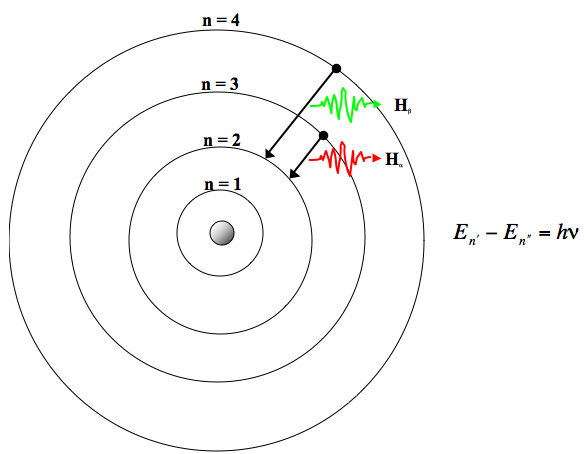
 The spin quantum number gives the spinning of an electron in a magnetic field and the wave characteristics of the electron.īohr model is an atomic model proposed by Niels Bohr (in 1915) to explain the structure of an atom. The magnetic quantum number describes the orientation of orbitals in the space. The angular momentum quantum number explains the shape of the orbital. The principle quantum number explains the average distance of the orbital from the nucleus and the energy level. Four types of quantum numbers are used in this The orbitals have particular shapes for different energy levels and are 3D structures.Īccording to the quantum model, an electron can be given a name with the use of quantum numbers. It also states that the orbitals are not always spherical. Thus, this model is mainly based on the probability of an electron to be located anywhere in the orbital. According to quantum theory, an electron has particle-wave duality and it is impossible to locate the exact position of the electron (uncertainty principle). This quantum model is based on quantum theory. Apart from that, Bohr used the Plank’s constant to calculate the energy of the energy levels of the atom.Īlthough the quantum model is much harder to understand than Bohr model, it accurately explains the observations regarding the large or complex atoms. Electrons can move from one energy level to another by absorbing or releasing energy.īohr model perfectly fit the hydrogen atom which has a single electron and a small positively charged nucleus.When an electron is moving in a certain orbital, the energy of that electron is constant. 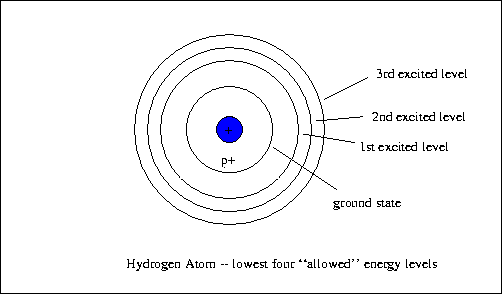
The atom is completely stable when electrons are in the lowest energy level.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
